 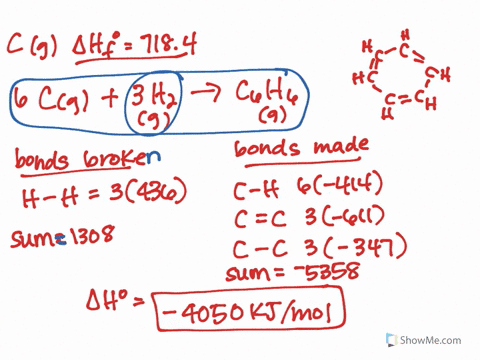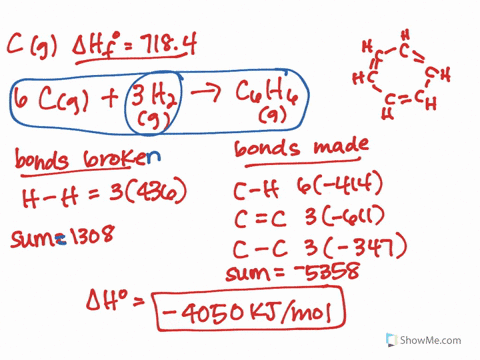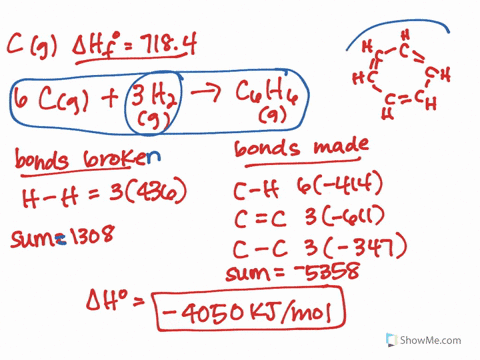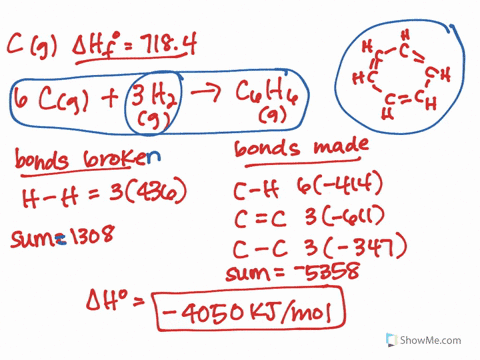 
It should follow from this that the lower-energy new eigenvector (bonding MO) will have a larger coefficient of the lower-energy old eigenvector (AO). The treatment would be similar to that which I showed here, except that you would need to explicitly calculate the actual eigenvectors. * This can all be formalised properly using quantum mechanics and linear algebra, but I won't do it here. In the case of HF and HI, fluorine has the highest electronegativity, which means the bond in HF is more polarised than in HI. Even, for example, carbon monoxide is problematic: it is only formed from two atoms, but its MOs generally have contributions from more than two different orbitals (carbon 2s and 2p, and oxygen 2s and 2p). It only truly works in the specific case where two AOs make up two MOs. Note that this can't be generalised to more complex molecules and MO diagrams. The electric dipole moment is a measure of the charge distribution in a molecule. This suggests that the contribution of the fluorine AO is larger often we say that the bonding MO is "centred on fluorine" or "localised on fluorine".* Effectively, the electrons in this MO would belong more to fluorine than they do to hydrogen. In this case, the bonding MO is closer in energy to the fluorine AO than it is to the hydrogen AO. Under these circumstances, it's possible to look at the energy levels to indirectly determine the contributions. In this case, however, the bonding MO (labelled $\sigma$) is only constructed from two constituent AOs: one from hydrogen ( $\mathrm_y$ AOs are nonbonding. This is actually not trivial to determine at all, and in general, the qualitative MO diagram you've attached isn't going to be sufficient. The key is to look at the occupied MOs and to determine whether they are mainly hydrogen-based, or fluorine-based (i.e., which atom contributes more to the MO). The compound that will have a permanent dipole moment. It's a bit more involved than that after all, the fluorine atom also has one of its electrons "moving down". 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
